
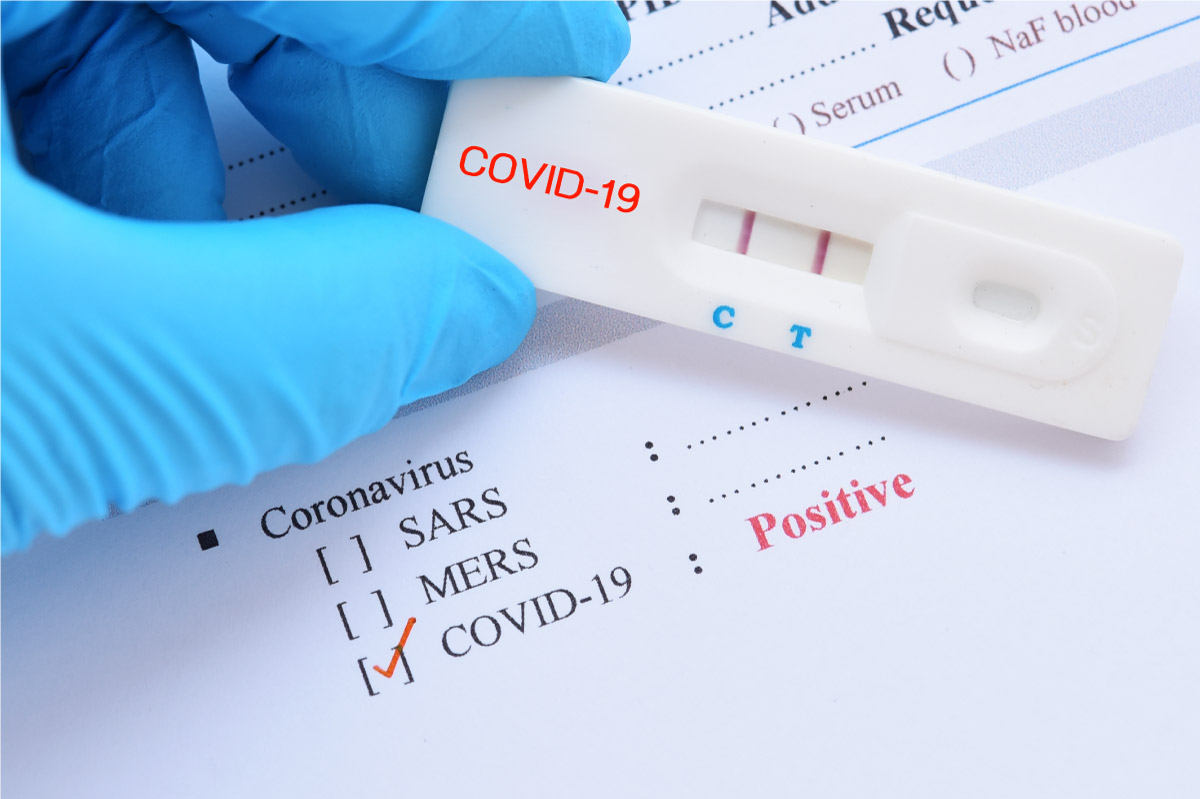
Acon FlowFlex Labeling Changes Mar 2023.FlowFlex COVID-19 Antigen Home Test Quick Reference Instructions.FlowFlex COVID-19 Antigen Home Test Package Insert for Healthcare Providers.FlowFlex COVID-19 Antigen Home Test Healthcare Providers Fact Sheet.FlowFlex COVID-19 Antigen Home Test Package Insert.FlowFlex COVID-19 Antigen Home Test FDA EUA Letter.Negative results should be considered in the context of an individual’s recent exposures, history and the presence of clinical signs and symptoms consistent with COVID-19Ĭlinical performance showed 93% sensitivity and 100% specificity Negative results are presumptive, and confirmation with a molecular assay, if necessary for patient management, may be performed Individuals who test positive should self-isolate and consult their healthcare provider as additional testing may be necessary and for public health reporting Positive results do not rule out bacterial infection or co-infection with other viruses Positive results indicate the presence of viral antigens, but clinical correlation with patient history and other diagnostic information is necessary to determine infection status This test is authorized for non-prescription home use with self-collected anterior nasal swab specimens directly from individuals aged 14 years and older or with adult-collected anterior nasal samples directly from individuals aged 2 years or older Product ships with minimum 60 days dating Qualitative detection of the nucleocapsid protein antigen from SARS-CoV-2 in anterior nasal swab specimens directly from individuals within 7 days of symptom onset or without symptoms or other epidemiological reasons to suspect COVID-19 infection Managing Global Supply Chain Challengesįlowflex Covid-19 Antigen Home test is for use under an FDA EUA:.Quick Results: Results in just 15 minutes!įlowFlex COVID-19 Antigen Rapid Test Kit - Individually PackagedĬall for bulk pricing discounts! 87 L031-118B5 EUA Number: EUA210494 This product is authorized by FDA under an Emergency Use Authorization.Molecular SARS-CoV-2 test (view in brochure) Accurate: Excellent performance when compared to an FDA authorized.Easy to use: Rapid Test Kit includes clear instructions to read results.Use with self-collected anterior nasal swab specimens directly from individuals aged 14 years and older or with adult-collected anterior nasal samplesĭirectly from individuals aged 2 years or older. The FlowFlex COVID-19 Antigen Rapid Test Kit test is authorized for non-prescription home Biological IndicatorsįlowFlex COVID Antigen Rapid Test Kit - Individually Packaged - Minimum order of 10 boxes (1 test per box) This is an at-home test kit with 15 minute results (no need to send to a lab) for the detection of COVID-19 antigens in nasal swab specimens. Sterilizer Monitoring results is minutes! 20 min.How To Wrap Instruments for RapidHeat Sterilization.AeroWipe™ Multipurpose Wipes and AeroCleanse™ Hand Sanitizer Moisturizing Gel. 
N95 Respirators Surgical Face Masks In Stock Same Day Shipping.Why Sterilizer Record Keeping Is Important.What is the difference between a Face Shield and a Face Mask?.Water Distillers - The Best Choice for Clean, Chemical & Mineral Free Water.SteriData Logger Paperless Sterilizer Cycle Printer.Patient recovery: The importance of human connection.NEW Instrument Washer Disinfector Steelco DS 500 CL.Is Your Instrument Washer Cleaning Effectively? Can You Prove It? Are Your Patients and Staff At Risk?.Introducing DuraCleanse Hands-Free Sanitizer Dispenser - Say No to leaky sanitizers and Save Money!.Infection Control: Sterilization Instrument Trays. 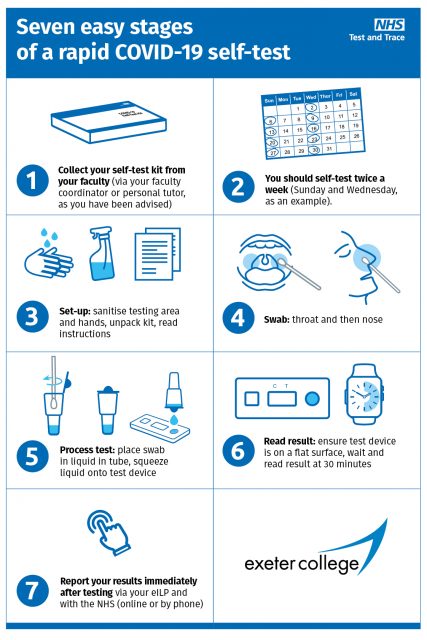
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
